Prevention
Modern medicine is increasingly transitioning towards preventive care. This shift towards prevention has also been observed in breast cancer care in recent years, particularly with the discovery of the BRCA gene. Subsequently, multiple genes and risk factors have been identified. Depending on these factors, a personalized screening strategy can be chosen. Therefore, it is crucial to understand these genetic and risk factors.
Diagnosis
I was diagnosed with cancer ... This website serves as a portal designed to assist you and your loved ones in accessing personal information and finding solutions to your concerns.
The primary goal of this website is to offer guidance and support to patients as they navigate their journey toward recovery and improved quality of life. The "Diagnosis" section of our website is divided into two main categories. Firstly, under "Anatomy and Physiology," we provide fundamental knowledge about the breast. Secondly, in the "Tumors and Disorders" section, we delve deeper into various breast-related conditions.
Moreover, we aim to provide information to women who may be concerned about potential breast issues but are hesitant to seek immediate medical advice. Knowledge and information can often offer immediate reassurance if a woman is able to identify the issue herself and determine that no specific treatment is necessary. Conversely, we also strive to educate women who have received a diagnosis of a serious breast condition, such as breast cancer, and wish to approach their doctor well-informed and prepared.
Treatment
The treatment for breast cancer should immediately include a discussion about reconstruction. Our foundation has no greater goal than to raise awareness of this among patients and oncological surgeons. By making an informed decision beforehand, we avoid closing off options for later reconstruction while still considering the oncological aspect. Of course, survival is paramount, and the decision of the oncologic surgeon will always take precedence.
The "Reconstruction or not?" page contains all the information you can expect during an initial consultation before undergoing tumor removal. This page is comprehensive, and your plastic surgeon will only provide information relevant to your situation.
"Removing the tumor" details the surgical procedure itself. This is the most crucial operation because effective tumor removal remains paramount. We guide you through the various methods of removal, a decision often made by a multidisciplinary team comprising oncologists, radiologists, pathologists, radiotherapists, breast nurses, gynecologists, oncological surgeons, and plastic surgeons.
The "Breast Reconstruction" section includes information and illustrations of the different reconstruction options along with corresponding steps.
Revalidation
Those treated for cancer often need a long period to recover.
Cancer is a radical illness with a heavy treatment. Often, people have to deal with psychosocial and/or physical problems afterwards, such as stress, anxiety, extreme fatigue, painful joints, reduced fitness, lymphedema... This can have a major impact on general well-being.
There are rehabilitation programmes offered by most hospitals. We cover some of the major topics here.
Quality of life
Quality of life is a key factor in coping with breast cancer. Therefore, it is important to find coping mechanisms that work, which will be different from patient to patient. For some, it may be finding enjoyment in activities they engaged in prior to diagnosis, taking time for appreciating life and expressing gratitude, volunteering, physical exercise... Of prime importance, studies have shown that accepting the disease as a part of one’s life is a key to effective coping, as well as focusing on mental strength to allow the patient to move on with life. In this section we are addressing some topics that patients experience during and after treatment and we are providing information to address them.
Tumors and diseases
Introduction
We have divides this part into infections, benign and malignant tumors.
Any new lump or change in the breast, nipple or armpit should be investigated.
The following methods of examination are commonly used:
Medical history and physical examination
Mammography, sometimes supplemented with ultrasound of the breast
Ultrasound of the breast distinguishes cystic from solid tumors and is the only diagnostic test suitable for adolescent girls.
Breast MRI (magnetic resonance imaging), in some instances an additional MRI of the breast is required. This may provide additional information about the nature of the lump, its location and extent.
Fine needle aspiration or tissue biopsy, possibly under ultrasound guidance. Under local anesthesia, a number of cells or a piece of tissue from the tumor are removed and sent for microscopic examination. This is usually sufficient to confirm the diagnosis of breast cancer.
For non-palpable tumors a stereotactic biopsy can be performed. Under ultrasound or MRI guidance, a guide-wire is placed within the tumor. This allows the surgeon to locate the tumor intra-operatively, where both the wire and surrounding tumor are removed.
Abnormal fluid discharge from the nipple can be investigated by galactography. A contrast medium is injected through a fine tube into the ducts, before performing a mammogram. This enables visualization and study of any abnormalities of the lactiferous ducts.
Discharge from the nipple can also be investigated by collecting some fluid on a glass plate, which can be examined for the presence of abnormal cells or blood.
I. Infections
Serious infections of the breast are unusual and the most common forms fall into the following categories:
Periductal mastitis
A fistula involving the breast or nipple
Puerperal mastitis
Other rare infections
A. Periductal mastitis
This results from a blockage of either secretions or milk, in the large ducts of the breast. It is currently unclear whether a blockage causes inflammation, or the inflammation occurs first, leading to duct obstruction. The fluid that builds up then becomes contaminated with bacteria, resulting in a localized infection. This causes pain, a breast mass, nipple discharge and ultimately a breast abscess. Frequently the classic signs of inflammation (redness, heat, swelling and pain) are all present.
Initial management involves a trial of conservative management with antibiotics. If a breast mass can be felt and is confirmed on ultrasound or mammography, then the mass should be biopsied. If there is any doubt, all suspicious masses should be excised.
B. Breast fistulas
A breast or nipple fistula is usually caused by a deeper abscess that opens into the nipple. Recurrent infections and fistulae are often seen in patients with inverted nipples. Chronic inflammation around the nipple also frequently leads to fistula formation.
Treatment involves incision of the fistula, identification of the abscess cavity, and then excision of both structures in-continuity. If any part of the abscess cavity or fistula remains, it is very likely that this will result in recurrence. A protracted period of wound care and antibiotics are sometimes needed.
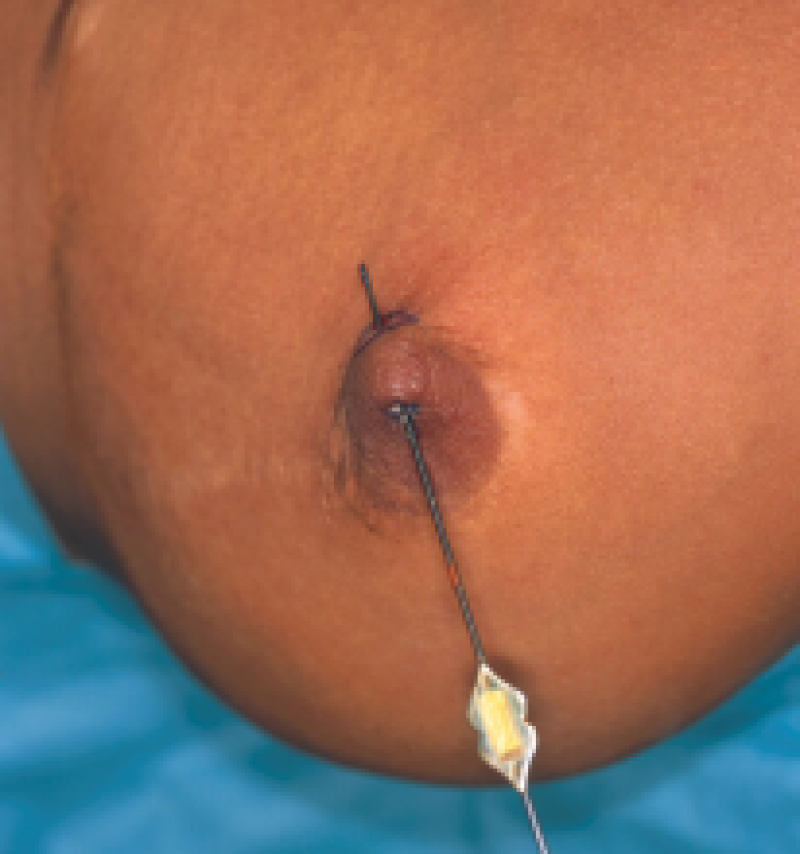
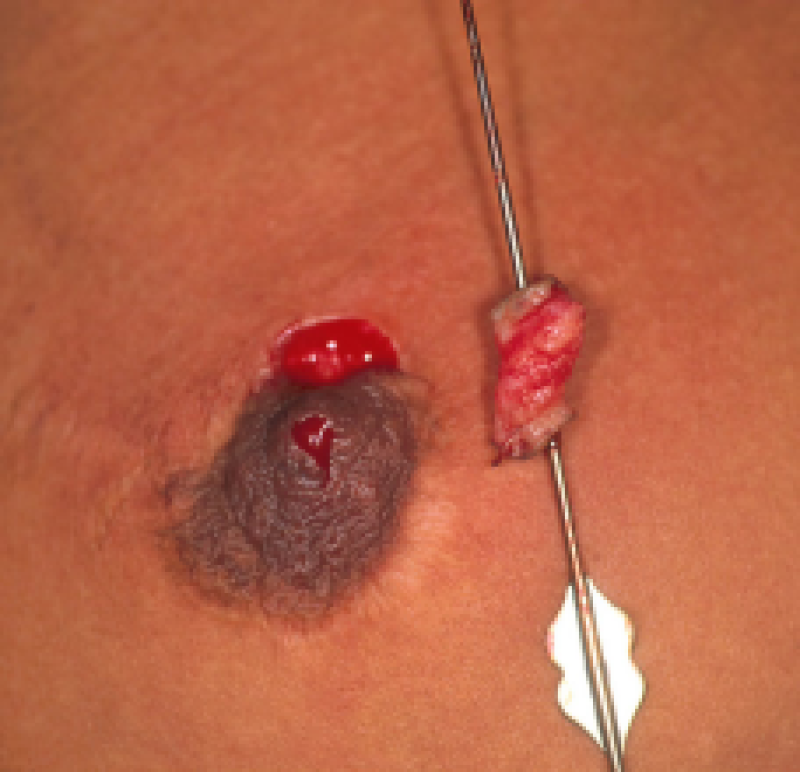
Figure: A chronic fistula diagnosed in a patient who had undergone a breast reduction, without correction of an inverted nipple.
Left: Exploration of the fistula under local anesthesia. Right: Complete excision of the fistula.
C. Puerperal mastitis
Following childbirth and during lactation, an area of adipose tissue around the mammary gland becomes inflamed. This is due to an acquired infection from either the newborn or a breast aspiration device. Typical symptoms include redness, heat, swelling and breast pain.
In most cases, treatment is conservative, with antibiotics. If the newborn baby is the source of the infection, it is sometimes necessary to discontinue breastfeeding until the symptoms resolve. In case of beginning mastitis, breast feeding may be continued but if you choose to stop breast feeding it is important to continue draining with a breast pump.
D. Other breast infections
Tuberculosis, syphilis, fungus and other rare pathogens occasionally cause severe breast infections. These can be difficult to diagnose, as they are seen so infrequently and mainly occur in underdeveloped countries. Specialized treatment is often necessary.
II. Benign Tumors
Benign tumors are local swellings in the breast. When such swellings are clinically and radiologically indistinguishable from a malignant tumor, they need to be surgically removed. Although a great variety of benign tumors exist, listed below are the most frequently encountered types:
A. Fibrocystic disease
These swellings develop as a result of fluid accumulation in small or large cysts (sacs). This condition occurs in about one third of women before the menopause. The typical complaint is of a swelling, sometimes painful, in the second half of the menstrual cycle. Sometimes the accumulations form microcysts, on other occasions you can clearly palpate the fluid filled cysts.
When the cysts are small, they decrease in volume after the return of menstruation. Large cysts (up to several centimeters in diameter) may persist. When clinically indicated, they may easily be drained under ultrasound guidance. If the wall of the cyst does not contain highly proliferating cells, this condition is not associated with any increased risk of breast cancer.
B. Duct ectasia of the breast
Here, milk secretion stops and causes inflammatory cells surrounding the ducts. There is an accumulation of concentrated material inside the ducts. This concentrated material can sometimes be seen beneath the nipple. After a while connective tissue forms, causing the ducts to contract, resulting in inflammation and pain.
C. Fibroadenoma
This is a common tumor. Fibroadenomas are firm, clearly defined swellings that are very mobile on manual examination. Surgery to remove them is relatively straight forwards. Fibroadenomas contain epithelial cells and connective tissue. They rarely grow fast.
D. Papilloma
These are benign tumors that grow in the breast gland ducts. They form around a central stalk with blood vessels. Epithelial cells proliferate around them. The solitary papillomas usually arise in one of the major ducts below the nipple complex. They may appear as multiple small papillomas in the depths of the breast tissue in smaller channels. Multiple papillomas have a slightly increased risk of malignant change.
E. Radial scar
Sometimes this can cause confusion. During mammographic screening, a radiologist may see a star-shaped lesion. It may resemble a malignant tumor with puckering. In reality, the star shaped strands are due to the connective tissue that surrounds the radial scar.
F. Benign microcalcification
These are small calcifications in the breast usually found incidentally on routine mammography. A radiologist is often able to determine whether or not these are benign. Microcalcifications occur as a result of stationary milk in the ducts that has calcified. If there is any doubt, any areas of calcification should be biopsied.
G. Fat necrosis
These are usually small, but can be large areas, where fat has died as a result of interruption of the local blood flow. Fat necrosis usually occurs after local trauma to the breast, for example, after a traffic accident.
III. Malignant tumors
A. Breast cancer risk factors
Breast cancer is the third most frequent tumor worldwide and the most common cancer in women. It accounts for 23% of all cancers in women.
In Belgium, about 1,500 women a year get breast cancer, worldwide the number is about 1.5 million. One woman in 9 will face the disease during her lifetime. The last 30 years have seen a growing incidence of breast cancer, but the good news is that the number of deaths due to breast cancer is decreasing. This is thanks to implemented screening and better treatment options.
Risk factors for developing breast cancer are:
Gender: Being female is the main risk factor, yet breast cancer can also occur in men. In 135 patients, there is 1 man with breast cancer.
Age: Breast cancer occurs mainly after the age of 55, but can also occur much earlier, starting at the age of 30-35. These very young patients are rather the exception.
Hormonal factors: In general, the longer the body is exposed to the influence of the female hormone estradiol, the higher the risk of breast cancer. This means:
An early puberty
a late menopause
a late first pregnancy or nullipare women
taking oral contraceptives: There is not sufficient evidence that the use of oral contraception would increase the risk of breast cancer. Overall, it can be assumed that use of the current, low-dose pill is safe, mainly if there is no increased familial risk of breast cancer.
Post-menopausal hormone replacement therapy: synthetic hormones will minimally increase the risk of breast cancer. Natural hormones, which we prescribe here in Flanders are breast safe.
prolonged breastfeeding would rather lower the risk
Pre-existing history of cancer: women with pre-existing ovarian cancer have an increased risk of developing breast cancer
Past history of breast cancer: women who have already experienced breast cancer have an increased risk of contralateral breast carcinoma. Thus, these women should be screened for life. Risk factors are: occurrence of breast cancer at a young age, familial mammary carcinoma, the presence of in situ cancers ( non-invasive cancers).
Ionizing rays: Exposure to ionizing radiation at a young age ( <30 years) or previous radiation for lymphoma, e.g. Hodgkin's disease.
Moderate alcohol consumption: Daily alcohol consumption can increase risk by 40%, but depends on family history and dietary factors.
Family history of breast cancer: 85% of patients have no family history of breast cancer. With disease of first-degree relatives ( mother, sister, daughter) the risk is significantly increased (2x). 5-10% of all breast cancers are hereditary. In these families breast cancer occurs very frequently, and often at a young age. Here a dominant hereditary factor can often be demonstrated (BRCA1, BRCA2 gene).
B. Examination
Through early detection, breast cancer can be recognized at an early stage. As a result, breast cancer mortality can be reduced. The following methods are used:
Breast self-examination: A significant proportion of breast cancers are diagnosed by the woman herself. Therefore, it is important that women themselves examine their breasts regularly. The best time for premenopausal women is 5-7 days after menstruation, for postmenopausal women a fixed day every month. How to do a breast examination yourself is explained in detail on the Internet
Clinical examination by a physician: From the age of 40, a clinical examination by a physician is recommended every 2 years.
Mammography: A screening mammogram can identify breast cancer at a very early stage, long before it becomes clinically palpable or visible. Obviously, smaller tumors have a much better prognosis. Moreover, treatment of very small tumors is often less invasive, and burdensome post-treatment with chemotherapy, for example, is not always necessary.
Recommendations for screening mammography:
around 40 years of age: baseline mammography
40-49 years: every year accompanied by a clinical examination by your physician
≥ 50 years of age: every year accompanied by a clinical examination by your physician, no age limit
C. Diagnosis
Any lump or change in the breast, nipple or armpit should be further examined. The following investigations are used for this purpose:
Firstly history and clinical examination
Mammography, usually supplemented by ultrasound of the breasts
Breast ultrasound: distinguishes cysts from tumours. Is usually the only examination in adolescent women.
MRI of the breasts (magnetic resonance imaging): When in doubt, an additional MRI of the breasts will be necessary in some cases. This examination can sometimes provide more information about the nature of the lesion, its localisation or extension.
Fine needle aspiration or tissue biopsy, possibly under ultrasound guidance: Under local anaesthesia, a number of cells or a tissue piece may be removed from the tumour, which will be further examined microscopically. This will usually allow the diagnosis of breast cancer to be confirmed.
For non-feeling tumours, a harpoon wire is placed under radiographic control. This harpoon remains in situ when the patient goes to the operating theatre, where the surgeon can remove the tumour with the harpoon wire.
In case of abnormal fluid loss from the nipple, a galactography will be performed. This involves the radiologist inserting contrast agent into the milk ducts through a fine tube, after which a mammogram is performed. This allows one to study the structure of the milk ducts and locate any abnormal milk ducts.
In case of loss of nipple fluid, some fluid can be collected on a glass slide, which is further examined for the presence of abnormal cells or blood.
D. Cancer types
When a breast glandular duct cell transforms into a malignant cell many things need to go wrong. Firstly, the duct cell needs to change its normal behavior and start dividing rapidly, but the cell also needs to survive. Most cells that transform at an abnormally high rate are disrupted by programmed cell death, called apoptosis. This programmed cell death does not occur in cancer cells however.
The wild, rapidly dividing cell, starts multiplying. The entire gland tube is filled with malignant cells and the lumen of the tube then disappears. As long as the cancer cells do not break through the wall or basal membrane of the duct, we refer to this cancer as a ‘carcinoma in situ’.
This is a non-invasive tumor with a good prognosis. The fact that the cells do not cross the wall of the basal membrane is crucial. The blood vessels and lymphatic vessels are located in the connective tissue around the gland tubes. As long as the cancer cells do not cross the basal membrane or wall of the gland tube, the cells cannot spread to other organs. The complete removal of a carcinoma in situ therefore guarantees a 100% cure.
Most non-invasive breast cancers do show an increased risk of developing an invasive component. It is therefore imperative that these non-invasive cancers are detected early and treated properly.
Once the cancer cells migrate through the basal membrane the cancer is called an invasive tumor. From now on the malignant cells can reach the blood and lymph vessels. The cells will form a mass outside the ducts that modifies the normal connective tissue distribution.
On examination of the breast a hard and immobile mass is felt. A malignant tumor is usually not painful. The tumor cells can migrate through the lymphatic vessels to the lymph nodes of the armpit or other areas around the chest. In patients with an invasive tumor, the sentinel node or the lower part of the axillary lymph nodes are removed during surgery and examined for metastases.
Non-invasive breast cancer
Ductal carcinoma in situ (DCIS) and lobular carcinoma in situ (LCIS) are noninvasive breast cancers. They are characterized by a proliferation of malignant cells in the mammary gland and ducts without penetration of the basal membrane.
DCIS is often diagnosed as microcalcification on mammography. DCIS is a precursor of invasive carcinoma and should always be fully removed. There are different degrees of aggressiveness, depending on:
The presence of necrosis
The grade of cell differentiation
The size of the tumor
The presence of microcalcification
LCIS is usually not palpable or visible on mammography. It is often an incidental finding in tissue removed for a different reason. LCIS is usually present in multiple places within the same breast and also both breasts may be affected. If LCIS is present, the risk of developing an invasive cancer is approximately 37%.
Invasive breast cancer
The cells of invasive cancers have the ability to invade and penetrate the surrounding tissues and can produce distant metastases in lymph nodes or other organs. The most frequent types are:
Invasive ductal carcinoma (IDC) (± 75%): This tumor usually presents as a hard mass in the breast and is often surrounded by DCIS.
Invasive lobular carcinoma (ILC) (± 5-10%): This often presents as a poorly defined thickening of the breast and is sometimes difficult to detect, as mammography and ultrasound can not always clearly highlight the tumor. In 90% of patients with ILC, LCIS is also found in the area. ILC is almost always highly hormone-sensitive and has an increased chance of developing in both breasts.
Less frequent types of invasive breast cancer include, medullary carcinoma, mucinous or colloid carcinoma and papillary carcinoma.
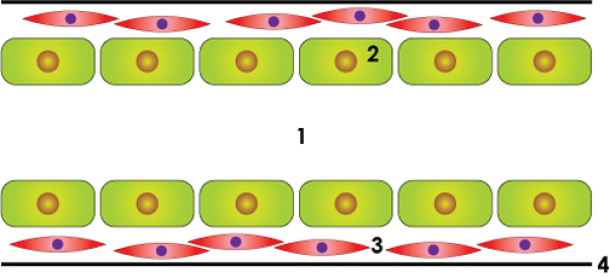
normal duct
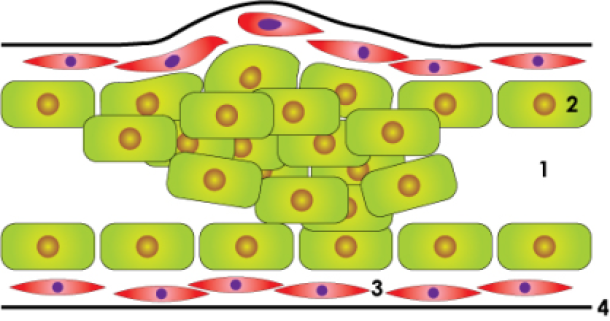
in situ carcinoma
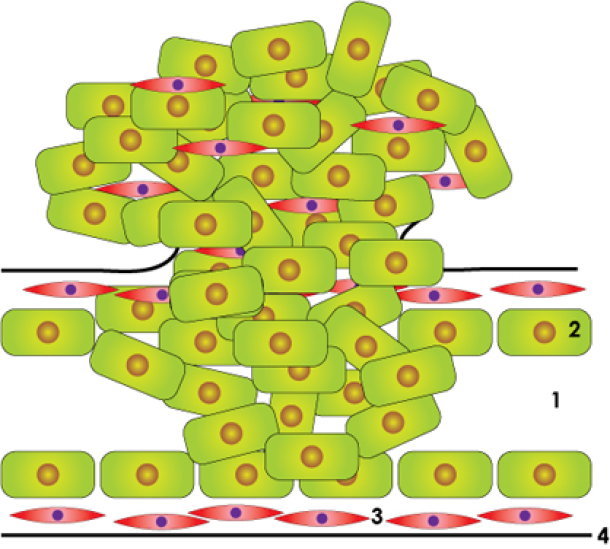
invasive cancer
Figure above: schematic drawing of a normal lactiferous duct, an in situ carcinoma and an invasive carcinoma: lumen (1), duct cells(2), smooth muscle cells (3), basal membrane (4).
Paget’s disease of the breast is a malignant condition that may have the appearance of eczema of the nipple, with skin changes involving the nipple and sometimes the skin of the breast. Since the condition in itself is often innocuous and limited to a surface appearance, it is sometimes dismissed, although actually indicative of a serious underlying cancerous condition.
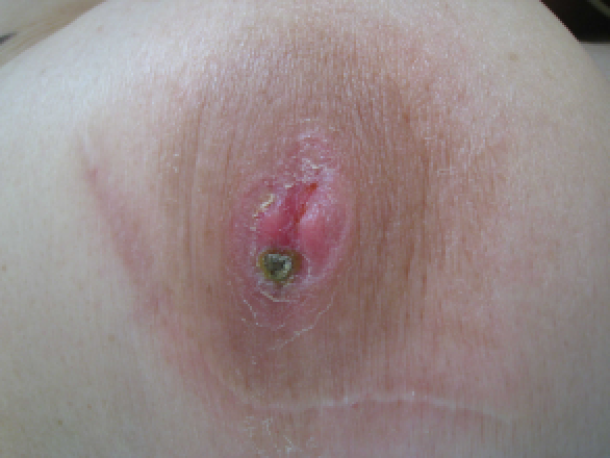
Paget's disease of the breast
E. Staging & prognosis
Breast cancer cells can spread through the body in the lymphatic system, the blood system or by direct growth into surrounding tissues.
The axillary lymph nodes are the first location where cancer cells may be held back, once they move from the primary breast tumor site. These lymph nodes are located in the armpit or along the chest wall lymphatics. From there cancer cells can spread into the venous system, and give rise to metastases in different organs: skeleton, lungs, liver, brain and soft tissues. Direct spread to the blood stream is also possible.
It is believed that breast cancer is often already a systemic disease at the time of diagnosis, meaning that there are already micrometastases throughout the body. Therefore there is a need to administer both locoregional treatment with surgery and radiotherapy, and adjuvant systemic therapy. This systemic treatment aims to destroy any micrometastases and thus avoid later relapse of the disease.
At the time of diagnosis of a breast cancer, proper staging is mandatory to determine the spread of the disease, the prognosis and the need for adjuvant treatment. The staging uses the TNM System, where T stands for tumor size, N for the status of the axillary nodes (whether or not they are affected), and M for the presence or absence of distant metastasis.
Tumor size (T)
Tumor size is correlated with prognosis (the larger the tumor, the worse the prognosis), but also with the status of the axillary nodes (the larger the tumor, the greater the chance of affected axillary nodes). Tumors of less than 1 cm usually have a good prognosis.
Lymph nodes status (N)
The axillary nodes are the primary, and usually first place of lymphatic drainage from the breast. The number of positive (or affected) axillary nodes is the main prognostic factor. The significance of so-called micrometastases in the axillary nodes (foci of tumor cells with a diameter <2 mm) is presently not well known. The table below shows the chances of developing a recurrence according to the status of the axillary lymph nodes.
N° of positive axillary nodes | Disease free survival at 5 years | Overall survival at 10 years |
0 | + 60% | 65-80% |
1-3 | 60-70% | 35-65% |
4 | + 56% | 15-25% |
>4 | 18-46% | 10-20% |
Presence or absence of distant metastasis (M)
If there are no proven metastases, adequate loco-regional treatment and drug therapy are curative. If metastases are detected in other organs, using conventional radiographic techniques, such as abdominal ultrasound and isotopic analysis of the skeleton, this means that the disease is incurable. The duration and quality of life can however be improved with different types of medication.
Prognostic factors are those factors at the time of diagnosis that give additional information about the potential outcome of the disease (risk of relapse, risk of death) and determine the need for adjuvant treatments.
The main prognostic factors are:
Histological grading, nuclear and histological grading: Grade I-II-III. The higher the grade, the poorer the differentiation and the worse the prognosis. This is determined by microscopic examination of the tissue removed.
Hormone Receptor Status (HR): Estrogen and progesterone receptors are proteins that are present on the membrane of the tumor cells. Blood hormones (estrogen or progesterone) bind to these proteins creating a sequence of signals that ultimately leads to cell division and thus, tumor growth. Hormone receptor-positive breast cancers have a better prognosis than hormone receptor-negative tumors. In additon, these receptors are a predictive factor for the success of an anti-hormonal therapy. This treatment blocks the stimulating effect of estrogen on cancer cells.
The presence of hormone receptors is determined immuno-histochemically and is expressed as a percentage of positive cells per existing cancer cells (0-100%). The degree of response to anti-hormonal treatment depends on the strength of presence of these receptors. About 60% of breast tumors are hormone receptor-positive.Her-2/neu receptor (c-erbB-2 receptor)The HER-2/neu or c-erbB-2 receptor is a protein present in the membrane of the tumor cells. When Growth Factors bind these proteins, a sequence of signals ultimately leads to cell division and thus, tumor growth. Overexpression (overactivity or a high number) of the HER-2 receptor is found in 20-25% of all breast cancers and is usually the result of amplification of the HER-2 oncogene. Overexpression of the HER-2 receptor is correlated with an aggressive cancer and a poor prognosis. It is also a predictive factor for response to a new “target” therapy with an antibody to the receptor, trastuzumab (Herceptin). Treatment with trastuzumab in HER-2 positive patients is useful in advanced stages of breast cancer and as adjuvant treatment. This treatment may decrease the risk of relapse by 50%.
Lymphatic and vascular invasion: the presence of tumor cells in the lymphatic vessels and blood vessels of the tumor or the presence of affected lymph nodes is a measure of aggressiveness and is a poor prognostic factor.
Age of patient: young patients often have poorly differentiated tumors with negative hormonal receptor status. Patients younger than 35 years are therefore regarded as high risk.
F. Adjuvant Therapy
In many cases additional treatment is required after surgery. This is called adjuvant therapy. The adjuvant treatment for breast cancer may include chemotherapy, hormonal therapy, radiotherapy or immunological therapy. Usually a combination of the above is administered. The objective of adjuvant treatment is to destroy residual cancer cells circulating in the body, preventing them from lodging in organs and giving rise to distant metastases. Currently there are no means to identify these circulating cancer cells, but based on a number of the characteristics of breast cancer the probability of recurrence or metastases can be estimated. From large international clinical studies, we know that chemotherapy and hormonal therapy can reduce the risk of relapse and death from breast cancer by 30-40%.
G. Follow-up
After completing the full treatment for breast cancer, careful follow-up is planned and performed to help early detection of any subsequent recurrence.
The following scheme is usually followed:
Years 1-2: every 3 months
Years 2-5: every 6 months
After 5 years: yearly appointment
The follow-up consists of a clinical examination and an annual mammogram. If necessary, blood samples and other radiological investigations may be requested.
In instances of local recurrence in the breast after breast-conserving surgery, a mastectomy can be performed with a good chance of cure.
The use of routine blood tests, chest radiography, liver ultrasound and bone scan is very controversial. Older clinical studies show no survival improvement for patients in whom these studies were regularly performed. This is partly explained by the lack of efficient treatment for breast cancer metastases.
Today new resources are available, and the survival of these patients is much improved. Therefore, a more detailed examination of some patients may be justified and necessary. Tumor marker detection in the blood is not helpful in screening for recurrence.
H. Local recurrence and metastases
Once metastases to other organs have occurred, definitive cure is unfortunately impossible. Hormone therapy or chemotherapy is then administered with the intention of reducing tumor volume and extending survival and improving patients quality of life.
Metastases from breast cancer can occur in all organs of the body. The most common sites are bone, liver, lungs, skin and lymph nodes. Extension of the disease can cause unpleasant symptoms such as pain, breathlessness, nausea, vomiting and weakness.
Treatments that reduce the tumor volume may result in a significant improvement of these symptoms, as well as extending survival.
Local recurrence in the breast does not necessarily mean an incurable situation as in this instance, a mastectomy can still be performed. If no metastases in other organs are identified, there is a real chance of a full recovery.
I. Male breast cancer
Although rare, breast cancer can also occur in men. Of all breast cancers, only 0.7% affect male patients, with an average age at diagnosis of 65 years. Risk factors include a history of undescended testis, infertility or rare hereditary disorders. Often male breast cancer presents in families confronted with hereditary breast cancer. Breast cancer in men is treated similarly to breast cancer in women.
To witness an example of male breast cancer, look at the following video:
